CAR-T
Treatment
Process
Individualized Treatment Specifically
for each Patient
CAR-T
Treatment
Process
Sub-Heading
CAR-T
Treatment
Process
Individualized Treatment Specifically for each Patient

-
1
Patient Eligibility and
Referral to a Certified Centre - 2
Apheresis - 3
Bridging Therapy - 4
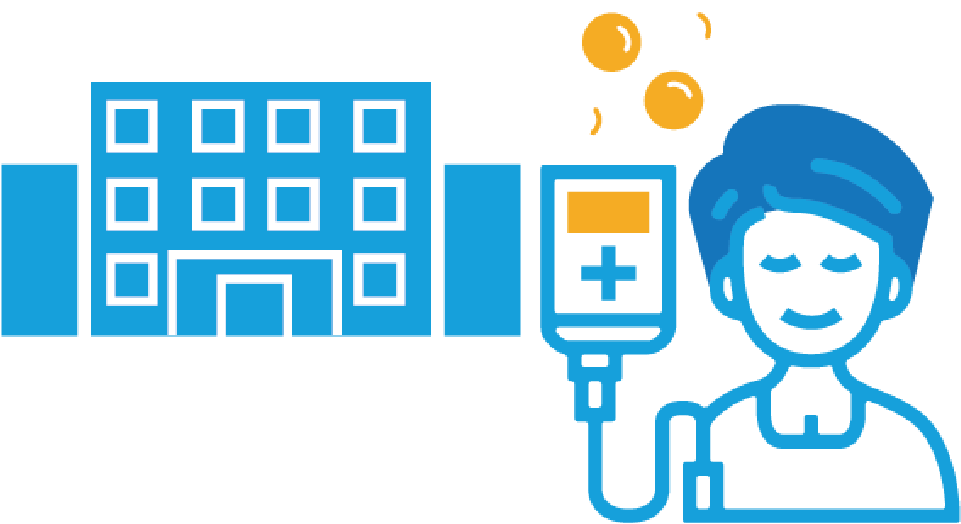
Lymphodepleting Therapy - 5
CAR-T Cell Infusion - 6
Short-term Monitoring - 7
Long-term Monitoring
Patient Eligibility and Referral to a Certified Centre
If you are a potential candidate for CAR-T Cell Therapy, you will be referred by a specialist to a certified CAR-T treatment centre. A panel of expert clinicians at the centre will then assess your candidacy for CAR-T Cell Therapy. If they confirm that you are eligible for treatment, they will schedule your appointment for the next stage in the process which is apheresis.

Apheresis
Apheresis will take approximately 3.5 – 4.5 hours*
*These timescales are approximate, and will vary between patients and treatment centres

Bridging Therapy
CAR-T cell manufacturing takes approximately 2 – 4 weeks*
*These timescales are approximate, and will vary between patients, treatment centres, the type of bridging therapy and the type of CAR-T Cell Therapy
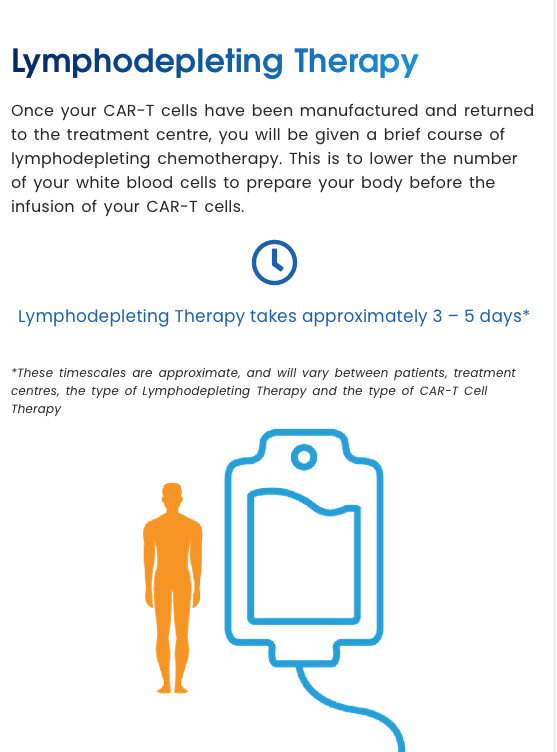
Lymphodepleting Therapy
Lymphodepleting Therapy takes approximately 3 – 5 days*
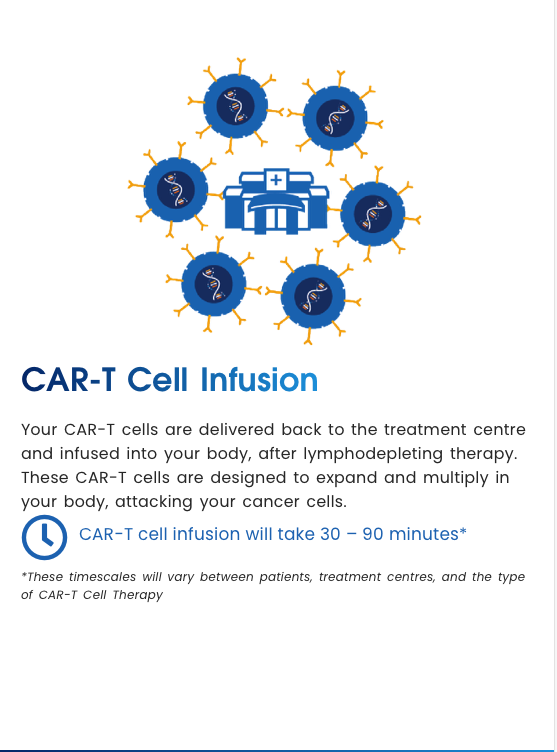
CAR-T Cell Infusion
CAR-T cell infusion will take 30 – 90 minutes*

Short-term Monitoring
You will need to stay within proximity of the treatment centre for at least 4 weeks after your CAR-T cell infusion.*

Long-term Monitoring
The timings of your long-term monitoring will depend on your clinical condition and individual needs, your healthcare team will discuss this with you.*


Long-term Monitoring
The timings of your long-term monitoring will depend on your clinical condition and individual needs, your healthcare team will discuss this with you.*
-
1
- 2
- 3
- 4
- 5
- 6
- 7
Patient Eligibility and Referral to a Certified Centre
If you are a potential candidate for CAR-T Cell Therapy, you will be referred by a specialist to a certified CAR-T treatment centre. A panel of expert clinicians at the centre will then assess your candidacy for CAR-T Cell Therapy. If they confirm that you are eligible for treatment, they will schedule your appointment for the next stage in the process which is apheresis.

Apheresis
Apheresis will take approximately 3.5 – 4.5 hours*
*These timescales are approximate, and will vary between patients and treatment centres

Bridging Therapy
CAR-T cell manufacturing takes approximately 2 – 4 weeks*
*These timescales are approximate, and will vary between patients, treatment centres, the type of bridging therapy and the type of CAR-T Cell Therapy

Lymphodepleting Therapy
Lymphodepleting Therapy takes approximately 3 – 5 days*

CAR-T Cell Infusion
CAR-T cell infusion will take 30 – 90 minutes*

Short-term Monitoring
You will need to stay within proximity of the treatment centre for at least 4 weeks after your CAR-T cell infusion.*

Long-term Monitoring
The timings of your long-term monitoring will depend on your clinical condition and individual needs, your healthcare team will discuss this with you.*


Long-term Monitoring
The timings of your long-term monitoring will depend on your clinical condition and individual needs, your healthcare team will discuss this with you.*
Variante 01
- 1 • Patient Eligibility
- 2 • Apheresis
- 3 • Bridging Therapy
- 4 • Lymphodepleting Therapy
- 5 • CAR-T Cell Infusion
- 6 • Short-term Monitoring
- 7 • Long-term Monitoring
Patient Eligibility and Referral to a Certified Centre
If you are a potential candidate for CAR-T Cell Therapy, you will be referred by a specialist to a certified CAR-T treatment centre. A panel of expert clinicians at the centre will then assess your candidacy for CAR-T Cell Therapy. If they confirm that you are eligible for treatment, they will schedule your appointment for the next stage in the process which is apheresis.

Apheresis
Apheresis will take approximately 3.5 – 4.5 hours*
*These timescales are approximate, and will vary between patients and treatment centres

Bridging Therapy
CAR-T cell manufacturing takes approximately 2 – 4 weeks*
*These timescales are approximate, and will vary between patients, treatment centres, the type of bridging therapy and the type of CAR-T Cell Therapy

Lymphodepleting Therapy
Lymphodepleting Therapy takes approximately 3 – 5 days*

CAR-T Cell Infusion
CAR-T cell infusion will take 30 – 90 minutes*

Short-term Monitoring
You will need to stay within proximity of the treatment centre for at least 4 weeks after your CAR-T cell infusion.*

Long-term Monitoring
The timings of your long-term monitoring will depend on your clinical condition and individual needs, your healthcare team will discuss this with you.*


Long-term Monitoring
The timings of your long-term monitoring will depend on your clinical condition and individual needs, your healthcare team will discuss this with you.*
Variante 02
The 7 Steps of the
CAR-T Process




Variante 03
The 7 Steps of the
CAR-T Process
Patient Eligibility and Referral to a Certified Centre
If you are a potential candidate for CAR-T Cell Therapy, you will be referred by a specialist to a certified CAR-T treatment centre. A panel of expert clinicians at the centre will then assess your candidacy for CAR-T Cell Therapy. If they confirm that you are eligible for treatment, they will schedule your appointment for the next stage in the process which is apheresis.

Apheresis
Apheresis will take approximately 3.5 – 4.5 hours*
*These timescales are approximate, and will vary between patients and treatment centres

Bridging Therapy
CAR-T cell manufacturing takes approximately 2 – 4 weeks*
*These timescales are approximate, and will vary between patients, treatment centres, the type of bridging therapy and the type of CAR-T Cell Therapy

Lymphodepleting Therapy
Lymphodepleting Therapy takes approximately 3 – 5 days*

CAR-T Cell Infusion
CAR-T cell infusion will take 30 – 90 minutes*

Short-term Monitoring
You will need to stay within proximity of the treatment centre for at least 4 weeks after your CAR-T cell infusion.*

Long-term Monitoring
The timings of your long-term monitoring will depend on your clinical condition and individual needs, your healthcare team will discuss this with you.*


Long-term Monitoring
The timings of your long-term monitoring will depend on your clinical condition and individual needs, your healthcare team will discuss this with you.*
1.
2.
3.
4.
Bo. Nam am eicte pro quunt omnime diosaer eptam, sus. Ibus, nonectae nos autatem int ressequi offic totat vel inulparum que nus. Niscianimo bla il.


 Bo. Nam am eicte pro quunt omnime diosaer eptam, sus. Ibus, nonectae nos autatem int ressequi offic totat vel inulparum que nus. Niscianimo bla il ea quo corum fugiant et et quatiundae eritatius vellum a cus aut labo. Nemporeicae quame mod magnati aspedi temporecto cus, ut est quis ium dolorpo ssequiatur mincilia eo
Bo. Nam am eicte pro quunt omnime diosaer eptam, sus. Ibus, nonectae nos autatem int ressequi offic totat vel inulparum que nus. Niscianimo bla il ea quo corum fugiant et et quatiundae eritatius vellum a cus aut labo. Nemporeicae quame mod magnati aspedi temporecto cus, ut est quis ium dolorpo ssequiatur mincilia eo

 Bo. Nam am eicte pro quunt omnime diosaer eptam, sus. Ibus, nonectae nos autatem int ressequi offic totat vel inulparum que nus. Niscianimo bla il ea quo corum fugiant et et quatiundae eritatius vellum a cus aut labo. Nemporeicae quame mod magnati aspedi temporecto cus, ut est quis ium dolorpo ssequiatur mincilia eos
Bo. Nam am eicte pro quunt omnime diosaer eptam, sus. Ibus, nonectae nos autatem int ressequi offic totat vel inulparum que nus. Niscianimo bla il ea quo corum fugiant et et quatiundae eritatius vellum a cus aut labo. Nemporeicae quame mod magnati aspedi temporecto cus, ut est quis ium dolorpo ssequiatur mincilia eos